Overview
ChemSee’s at-home detectors utilize well-known laboratory techniques for the detection of poisons in foods. These state-of-the-art chemistries are packaged together in small, easy-to-use formats which easily can be used by any person can easily use to test their food immediately before eating. ChemSee’s detectors were developed in cooperation with the Department of Defense to ensure accuracy and proper / meaningful results.
Available Products
Detectors For Food Contaminants
Available Products for the Detection of Adulterants in Food, Drinks or Water including:
Peroxide, Borax, Nitrates and Nitrites.
In-Lab Sample Testing
Testing is available for the detection of:
- Common Poisons (30+ Poisons including Cyanide, Azide, Heavy Metals and Arsenic)
- Ethylene Glycol (Antifreeze)
- Other
Antifreeze Detection

Detectors that allow the user to rapidly check if a suspicious sample of food or drink contains ethylene glycol / antifreeze.
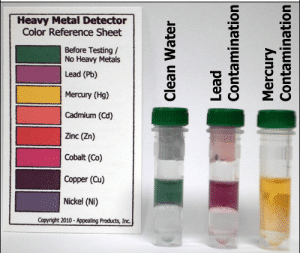
Heavy Metal Detectors
Easy-to-use detectors to instantly detect trace levels of Heavy Metals in foods or liquids.
Detectors for Sulfites
Sulfites are used to preserve foods and help them keep their color. Sulfites are also toxic and can cause life-threatening conditions to people whom have Asthma or an allergic condition.
What Are ChemSee’s Tests?
ChemSee’s Detectors for Foods are a class of rapid, presumptive tests that can indicate on the spot if there is a reasonable probability that the food is contaminated.
ChemSee offers detectors to improve food safety in two ways:
- Rapid tests for determining if particular food contaminants are present, and,
- Presumptive tests of products for specific contaminants.
ChemSee is pleased to provide innovative products to ensure both the safety and quality of foods for the consumer. From the innovative Milk-Ox Detectors for peroxide, to SulfiteCheck detectors for sulfites in foods and drink, ChemSee is committed to providing the tools necessary to ensure that your food is safe.